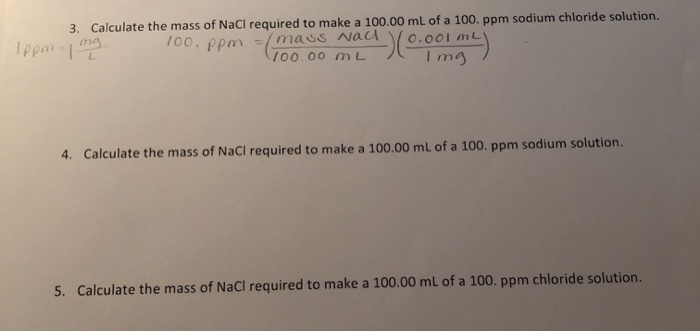
- How to calculate ppm by mass how to#
- How to calculate ppm by mass full#
- How to calculate ppm by mass plus#
While ppm is the volume/volume ratio or the mass/mass ratio, mg/L is the mass/volume ratio. The unit of PPM can be converted to mg/L by multiplying by. One part per million equals one milligram per liter. A reading of 6 mg/L corresponds to 6 ppm or 6000 ppb, which corresponds to 6000 g/L. 1 ppm = mg/lįor water, 1 ppm = approximately 1 mg/L (also called mg/L) of contaminants in the water and 1 ppb = 1 μg/L (also called g/L). For example, for chloride, the molar mass of a solution with a concentration of 1 M has g chloride per 1 liter of solution. To convert molarity to ppm, you must first determine the molar mass of the substance. Divide the mass of the first solid in milligrams by the mass of the second solid in kilograms. To calculate the PPM of one solid mixed with another, compare the masses of the two. That is, ppm = (mass of solute ÷ mass of solution) x 1,000,000. The concentration in parts per million or ppm is very similar to the weight percentage, except it multiplies the mass ratio by 1,000,000 instead of 100. How do you calculate the concentration of ppm?
How to calculate ppm by mass how to#
How to convert a number from percent (%) to parts per million (ppm). This number is then multiplied by 106 and expressed in parts per million (ppm). To find parts per million, divide the mass of the solute by the total mass of the solution. What is the formula for parts per million? Divide the mass of the solute by the total mass of the solution.
How to calculate ppm by mass plus#
The total mass of the solution is equal to the mass of the solvent plus the mass of the solute. Find the total mass of the solution in g. Measure the mass of the solute you want to mix with your solution. Method 2 of 3: Find the concentration in percent or parts per million. How they can calculate the concentration in ppm?
+Convert+mass%2Fvolume+to+mass%2Fmass.jpg)
So one part per million is a percentage: 1 ppm = To convert ppm to percent, divide ppm by 10,000: x (%) = x (ppm) / 10,000. How to Convert PPM to Percentage How to Convert Parts Per Million (ppm) to Percentage (%). 1 kmole SO 4 2- is 2 keq, (because of the double - charge) and 1 keq SO4= is 32/2 or 16 kg S.What does 1 ppm equal? One ppm is equal to 1 milligram of something per liter of water (mg/L) or 1 milligram of something per kilogram of soil (mg/kg). The 'equivalent' refers to the ionic charge on an ion.
How to calculate ppm by mass full#
The full process is to convert kg to kmoles, then kmoles to keq. For sulphur, the prevalent ion is sulphate SO 4 2-, which has a double charge (valency). For example NH 4 + or NO 3 - have a valency of 1. When converting acidifying compounds the valency is important and is connected with the charge on the ion. Kg ha -1 year -1 to kiloequivalents ha -1 year -1: Their concentrations are normally expressed only in µg m -3 sulphate, nitrate) are present as particles in theĪir and the concept of a volume mixing ratio of gases is not obviously applicable. Molecules and has a partial pressure of one millionth of the atmosphericĮquates to a different air concentration in µg m -3įor this reason air concentrations of NO X are normally expressed as µg NO X-NO 2 Of polluted air, it is also 1 molecule per 10 -6 As a consequence of the gas laws, a gas presentĪt a volume mixing ratio of 1 ppm is not only 1 cm 3 Upon temperature and pressure as these affect both the pollutant and theĪir to the same extent. Ideal gas behaviour is assumed and thus the concentration is not dependent Volume if segregated pure, to the volume of the air in which it is contained.

This unit expressed the concentration of a pollutant as the ratio of its Volume mixing ratio: usually ppm - parts per million (10 -6) Units should, strictly speaking, specify the conditions at the time of sampling. Of a given parcel of air is dependent upon the temperature and pressureĪt the time of sampling, the pollutant concentration expressed in these

Pollutant is expressed as a ratio to the volume of air. There are two systems of unit in common use for gaseous and particular pollutants: Mass per unit volume: usually µg -3.
